
Isotopes are discussed more in depth later in this module. Since isotopes have a different number of neutrons, their mass numbers and atomic masses differ from those listed in the periodic table. The atomic mass is typically listed in the periodic table below the element's name. The atomic mass of Hydrogen is 1.00797 amu and the atomic mass of Carbon is 12.011 amu. The mass number is approximately equal to the atomic mass, which is the mass of a single atom of a element measured in atomic mass units (amu). 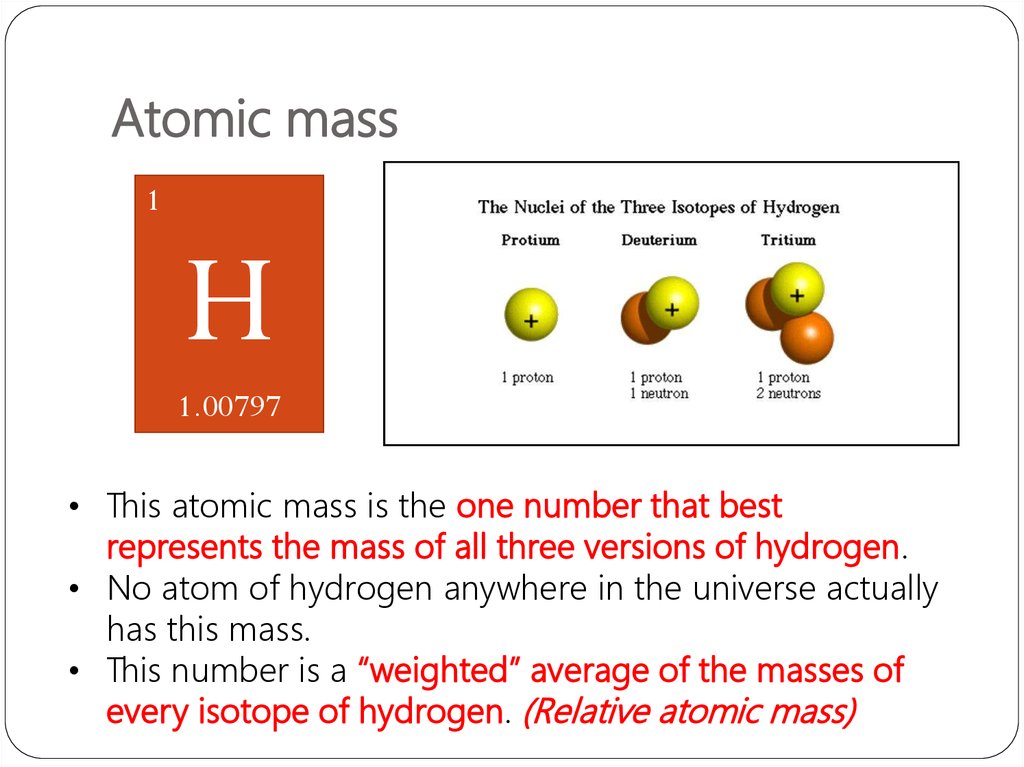
For example the mass number of a regular carbon atom is 12, since a carbon atom has 6 protons and 6 neutrons in its nuclus. In words, the mass number is the number of neutrons in an atom of a specific element plus the number of protons in an atom of that element. Isotopes are forms of elements that have the same number of protons and therefore the same atomic number, but a different number of neutrons which affects their mass number. Mass NumberĪll atoms have a mass number which is derived as follows: The relationship between the mass m and the molecular weight M M is the following: n m M M m n ×M M, where, n is the number of mole of the substance in question. The atomic number of an element never changes, meaning that the number of protons in the nucleus of every atom in an element is always the same. Explanation: The density D is calculated by: D m V. Oxygen atoms contain 8 protons and have an atomic number of 8. 
Atomic mass is sometimes erroneously confused with atomic weight the obsolete term for relative atomic mass. 1 atomic mass unit is defined as 1/12 of the mass of a single carbon-12 atom.:18 1 u has a value of 1.660 539 066 60(50) × 10 27 kg. Instead, atomic mass is expressed in unified atomic mass units (unit symbol: u). All carbon atoms, and only carbon atoms, contain six protons and have an atomic number of 6. The atomic mass of an atom (i.e., a specific isotope of an element) is measured in comparison with the mass of one atom of carbon-12 ( 12 C) that is assigned a mass of 12 atomic mass units (amu). However, because each atom has a very small mass, this is not easy to do. For example, all hydrogen atoms, and only hydrogen atoms, contain one proton and have an atomic number of 1. In other words, each element has a unique number that identifies how many protons are in one atom of that element. The number of protons in the nucleus of an atom determines an element's atomic number. Each proton or neutron weighs about 1 amu, and thus the atomic. Define and determine the mass number of an atom. Most of the mass of an atom is concentrated in the protons and neutrons contained in the nucleus.Define and determine the atomic number of an atom.As mentioned in the sections on protons and neutrons, both of these particles have. After reading this section you will be able to do the following: Atomic mass is exactly what it sounds like- a measure of the mass of an atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
